Pipeline
A comprehensive and differentiated cell therapy pipeline
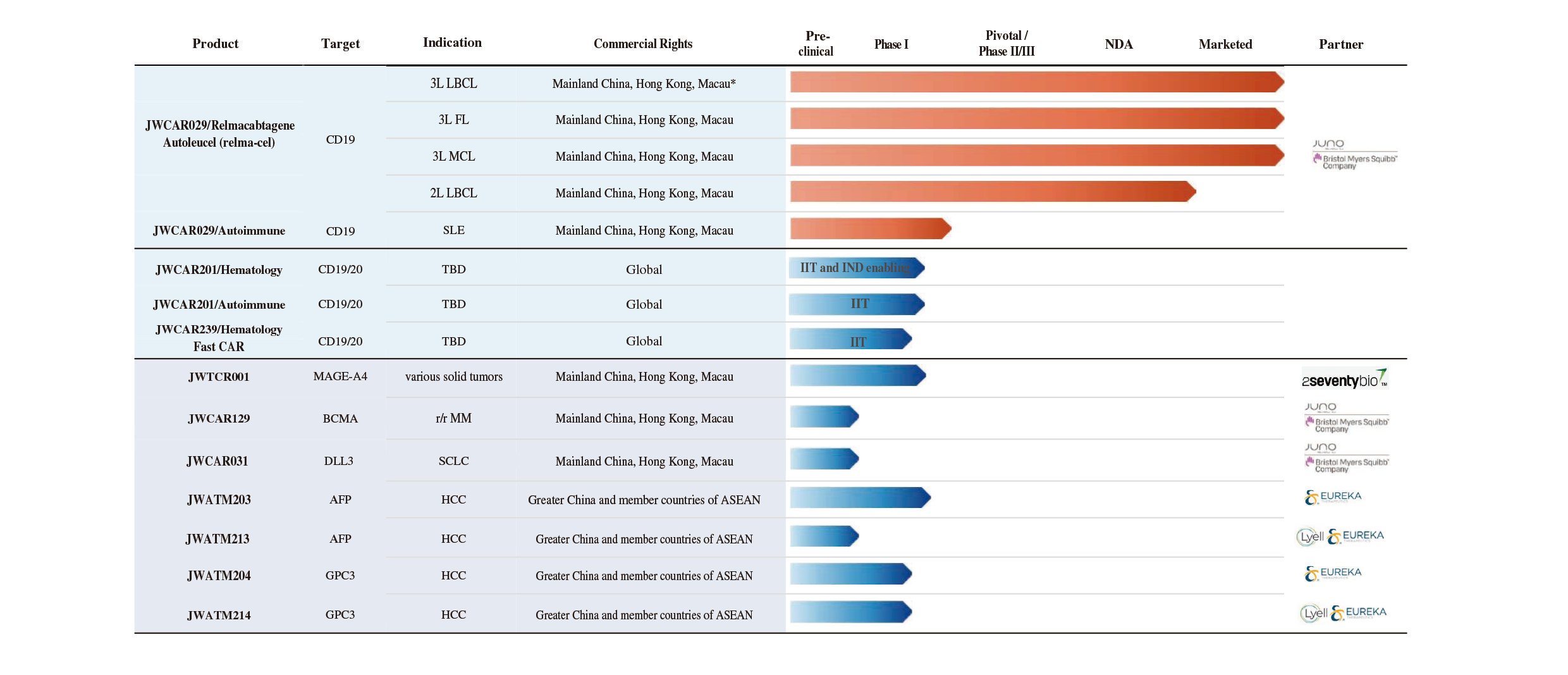
Information as of: Mar 2026
Abbreviations: LBCL = large B-cell lymphoma; FL = follicular lymphoma; MCL = mantle cell lymphoma;
SLE = systemic lupus erythematosus; r/r = relapsed or refractory; MM = multiple myeloma; SCLC = small cell lung cancer; HCC = hepatocellular carcinoma; MAGE-A4 = melanoma associated antigen A4; DLL3 = Delta-like ligand; AFP = alpha-fetoprotein; GPC3 =glypican-3.
* Mainland China, Hong Kong, Macau refer to Mainland China, Hong Kong (China), Macau (China), respectively.
1.Relma-cel is based on the same chimeric antigen receptor (“CAR”) construct as the product lisocabtagene maraleucel (Breyanzi or lisocabtagene or liso-cel) of Juno, which was approved by the U.S. Food and Drug Administration (“FDA”) in February 2021.
2.JWCAR129 is based on the same CAR construct as Juno’s product orvacabtagene autoleucel (orvacel).
3.SLE is a chronic autoimmune disease characterized by the production of autoantibodies and abnormal B-lymphocyte function.
4.JWATM204 is in a Phase I investigator-initiated trial in China. Eureka’s products based on the CAR constructs underlying JWATM203 and JWATM204 are currently in Phase I/II trials in the US conducted by Eureka under an IND application. In November 2021, the FDA granted Fast Track
Designation to Eureka’s counterpart to JWATM203 for the treatment of hepatoblastoma (“HB”) and HCC in pediatric patients, as well as “rare pediatric disease designation” for the treatment of HB. In February 2022, the FDA granted Orphan Drug Designation to Eureka’s counterparts to JWATM203 and JWATM204.
5.JWATM214 is developing using Lyell technology.
